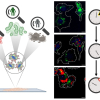
Long cycle life and high levels of safety are required for the large-scale application of energy storage devices (ESDs). It is important to understand both the operating and failure mechanisms of ESDs. Previously used characterisation techniques, such as X-ray diffraction (XRD), transmission electron microscopy, X-ray spectroscopy and topography, and nuclear magnetic resonance, are based on bulk regions of electrodes or electrolytes, and they have overlooked the critical surface/interface behaviours that govern the operation and failure in ESDs.
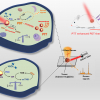
Recently, a research team led by Professor Fu Qiang from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) revealed the atmosphere-dependent relaxation and failure mechanisms of ESDs by in situ Raman spectroscopy, XRD and X-ray photoelectron spectroscopy (XPS).
They found that, for an aluminium ion battery, the relaxation effects of the graphite electrode in anhydrous atmospheres were manifested by recoverable stage–structure change and electronic relaxation. The mechanism could be described as the redistribution of the anion/cation pairs within the graphite electrode by in situ XPS. Once exposed to hydrous atmospheres, H2O molecules from ambient could intercalate into the graphite electrode and hydrolysis reactions could be induced between the newly intercalated H2O and ions. After H2O intercalation and hydrolysis, the failure behaviours of the graphite electrode occurred as shown by the stage–structure degradation and electronic decoupling.
“We have developed the atmosphere-, temperature- and potential-controlled operando/in situ surface/interface techniques and well-defined model devices”, said Professor Fu. “Such methods can be extended to explore the relaxation and failure mechanisms of more ESDs, such as metal–ion secondary batteries/supercapacitors, and the interface reactions in metal–gas batteries.”