Ingeborg E. Iping Petterson and Freek Ariese
LaserLaB, VU University Amsterdam, De Boelelaan 1083, 1081 HV Amsterdam, The Netherlands
Introduction
Raman spectroscopy is an increasingly important analytical tool, enabling fast, non-invasive, identification based on molecular vibrations. However, in the case of opaque materials, it is not possible to focus on deeper layers and in conventional Raman spectroscopic measurements the spectra are dominated by signals from the surface. To overcome this, advanced Raman spectroscopy instruments are being developed that rely on the principle that surface Raman photons and Raman photons from deeper within a sample can be discriminated on the basis of their distribution in time or space as they travel through an opaque material and are diffusely scattered and delayed.
Spatially offset Raman spectroscopy (SORS)1 makes this discrimination spatially; one can improve the ratio of deeper-to-surface Raman photons reaching the detector by exciting the sample at a different location from where the Raman signal is collected. Alternatively, time-resolved Raman spectroscopy (TRRS) techniques rely on short laser pulses and gated detection to make the discrimination; deeper Raman photons arrive at the detector later and one can selectively collect these photons by delaying the opening of a short detector gate. The relatively short gate width used in TRRS also considerably reduces fluorescence. We found that TRRS measurements, using an intensified charge-coupled device (ICCD) detector to create a gate of 250 ps in the case of our setup, provides better selectivity over SORS in subsurface measurements on a mm–cm depth scale.2
The ability to obtain chemical information through opaque, diffusely-scattering layers shows particular promise for a number of fields, including forensics and security screening and holds great potential for biomedical applications. Using this technique, we have detected explosives through white plastic containers and performed measurements through an artificial skin material, as well as through porcine skin in vitro. Also, preliminary data indicate that this technique has potential for use as an analytical tool in monitoring catalytic reactions in situ within catalytic extrudates.
Instrumentation
Our excitation source is a tuneable, pulsed Ti-Sapphire laser (Coherent Mira 900P, 3 ps pulses 76 MHz) pumped by a frequency doubled Nd:YVO4 laser (operated at 12 W, 532 nm; Coherent type Verdi-V18, Santa Clara, CA, USA), as seen in Figure 1. The pulse width of 3 ps is an important parameter; longer (ns) pulses would not result in sufficient temporal resolution, whereas shorter (fs) pulses would induce spectral broadening. The skin measurements were performed using 720 nm excitation, while the explosives and catalytic measurements were performed at 460 nm excitation after frequency doubling the Ti-Sapphire output.

Time-resolved measurements were carried out via an intensified CCD camera (LaVision Picostar, Göttingen, Germany) with a multichannel plate (MCP) operated at 750–850 V, (T = –11°C). The optimal gate width is typically 250 ps FWHM.
This setup can therefore be used for TRRS and SORS combined: TR-SORS. A spatial offset can be created by laterally moving the position of a small prism that directs the excitation beam onto the sample, while the collection optics remain static (see Figure 1). For non-gated SORS measurements, the CCD camera (CCD2, mounted on the same spectrograph; Andor Technology, Model DV 420-OE, Belfast, UK) can also be used.
Applications
Non-invasive explosives detection
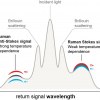
Using the TRRS technique, we were able to detect the Raman signals of various components of explosives, including 2,4- and 2,6-dinitrotoluene (DNT), akardite II, diphenylamine and ethyl centralite, through common plastic packaging materials of different thicknesses.3 Figure 2 depicts spectra of a sample of 2,4-DNT placed behind a 5 mm thick layer of polyethylene (PE). A depth profile of this sample was obtained by performing a series of measurements in which the delay time was increased in steps of 50–100 ps. We were able to measure a clean spectrum of the first layer of material (plastic) at a delay time of 0 ps and a longer delay time (300 ps) provided a spectrum of mixed signals from the first and second layers, from which we could then subtract the scaled first layer spectrum. The result was a relatively clean spectrum of the second layer of material (Figure 2). The ability to cleanly distinguish first and second layers using this technique has advantages over SORS for the identification of unknowns.

Through-skin measurements
In our on-going research into the application of this technique for biomedical diagnosis, we have performed a depth profile series of sample systems using artificial optical skin phantoms4 (OSPs) or porcine skin in vitro as first layer materials.
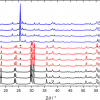
Using the TRRS technique, we were able to perform depth measurements of bulk reference materials, including a block of polyethylene terephthalate (PET) as shown in Figure 3, through several mm of OSP or porcine skin. We found that OSPs with a concentration of 0.4% TiO2 particles suspended in silicone (wt/wt) [Figure 3(a)] showed photon-scattering properties similar to those of porcine skin of the same thickness [Figure 3(b)]; a result that agrees well with optical coherence tomography studies of this material by de Bruin et al.4 Note, for both samples, the first appearance of the second layer peaks (PET) occurs at 100 ps and then these increase in intensity with longer delay times.

Depth analysis in catalytic extrudates
We modelled a catalytic reaction and were able to obtain depth-selective chemical information non-invasively from within real catalytic extrudates by TRRS (unpublished results). The use of Raman spectroscopy for non-invasive chemical analysis is not unknown in catalysis, but fluorescence often remains a problem. Our preliminary results indicate that the TRRS technique has great potential in this field and we found that this approach provided a better fluorescence reduction than SORS measurements in subsurface studies of these samples. Our on-going measurements in this topic include using different model samples and providing selective enhancement of particular chemical components within the catalytic pellet via surface enhanced Raman spectroscopy by the introduction of metal nanoparticles.5
Conclusion
Both TRRS and SORS enable one to selectively probe at depth within opaque samples. SORS can be carried out with continuous wave excitation and detection and is, therefore, currently more affordable and closer to commercial application (for example, those developed by Cobalt Light Systems, Oxford, UK). TRRS requires more complex instrumentation, but has specific advantages for difficult analytical problems. For example, fluorescence interference is generally better reduced by time gating. A unique feature of our setup is the ability to combine or compare TRRS with SORS on the same sample under the same excitation conditions. In addition, the advantage of a tuneable laser system is that it is possible to tailor the excitation wavelength to best suit the samples one would like to investigate. This will be particularly important for resonance Raman spectroscopy and for strongly absorbing or fluorescent signals.
The versatility of this home-built system allows for many different varieties of Raman spectroscopic measurements and in this overview we have discussed applications of this system in proof-of-principle experiments that are relevant to security, biomedicine and catalysis. We hope to continue to improve on this system and develop increasingly realistic applications in these and other fields.
Acknowledgements
We would also like to thank Cees Gooijer and Joost B. Buijs for their scientific and technical input. We would also like to thank María López-López and Carmen García-Ruiz (University of Alcalá, Spain) for their collaboration on the explosives detection work and the Government of Spain (Project CTQ2008-00633-E) and LaserLaB Europe (EU-Integrated Infrastructures Initiative Program No. 2008-1-228334) for making the collaboration possible. Thanks to Maarten Fokker for his work on the porcine skin measurements and to D. Martijn de Bruin at the Amsterdam Medical Center for providing OSP recipes and access to the OSP lab. Thanks to Clare Harvey, Arjan Mank, and Bert Weckhuysen (Utrecht University) for their collaborative work on the catalysis analysis.
References
- P. Matousek, I.P. Clark, E.R.C. Draper, M.D. Morris, A.E. Goodship, N. Everall, M. Towrie, W.F. Finney and A.W. Parker, “Subsurface probing in diffusely scattering media using spatially offset Raman spectroscopy”, Appl. Spectrosc. 59, 393–400 (2005). doi: 10.1366/0003702053641450
- I.E. Iping Petterson, P. Dvorak, J.B. Buijs, C. Gooijer and F. Ariese, “Time-resolved spatially offset Raman spectroscopy for depth analysis in diffusely scattering layers”, Analyst 135, 3255–3259 (2010). doi: 10.1039/c0an00611d
- I.E. Iping Petterson, M. Lopez-Lopez, C. Garcia-Ruiz, C. Gooijer, J.B. Buijs and F. Ariese, “Noninvasive detection of concealed explosives: Depth profiling through opaque plastics by time-resolved Raman spectroscopy”, Anal. Chem. 83, 8517–8523 (2011). doi: 10.1021/ac2018102
- D.M. de Bruin, R.H. Bremmer, V.M. Kodach, R. de Kinkelder, J. van Marle, T.G. van Leeuwen and D.J. Faber, “Optical phantoms of varying geometry based on thin building blocks with controlled optical properties”, J. Biomed. Opt. 15, 025001 (2010). doi: 10.1117/1.3369003
- C. Harvey, I.E. Iping Petterson, J.B. Buijs, F. Ariese, B.M. Weckhuysen, C. Gooijer and A. Mank, “Looking inside catalytic extrudates with time-resolved surface enhanced Raman spectroscopy; manuscript in preparation.